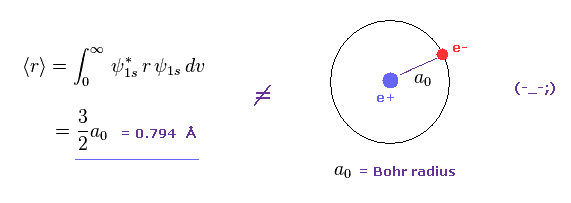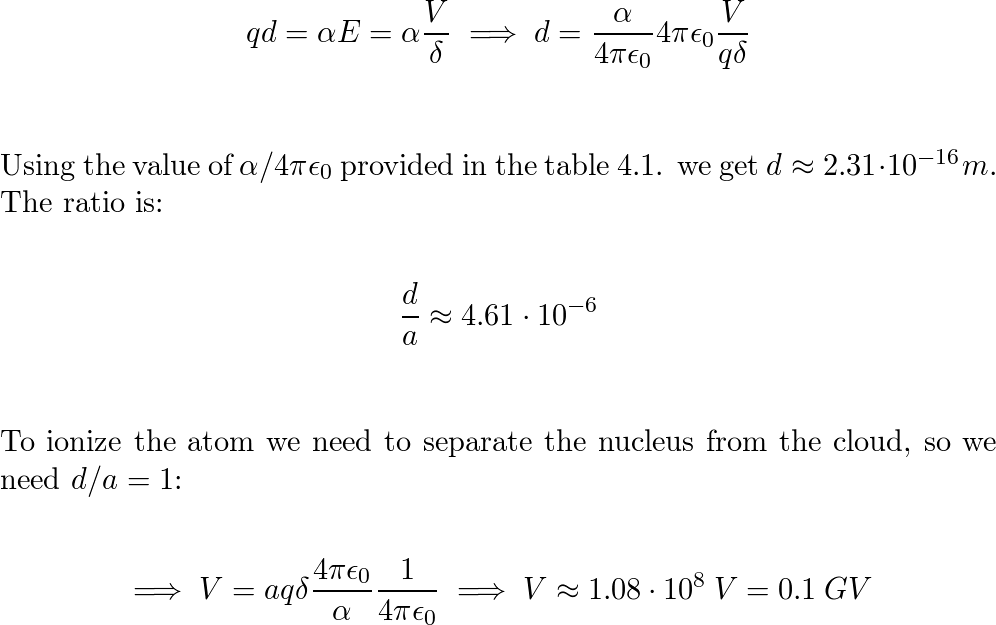SOLVED: The Bohr radius a0 is the most probable distance between the proton and the electron in the Hydrogen atom, when the Hydrogen atom is in the ground state The value of
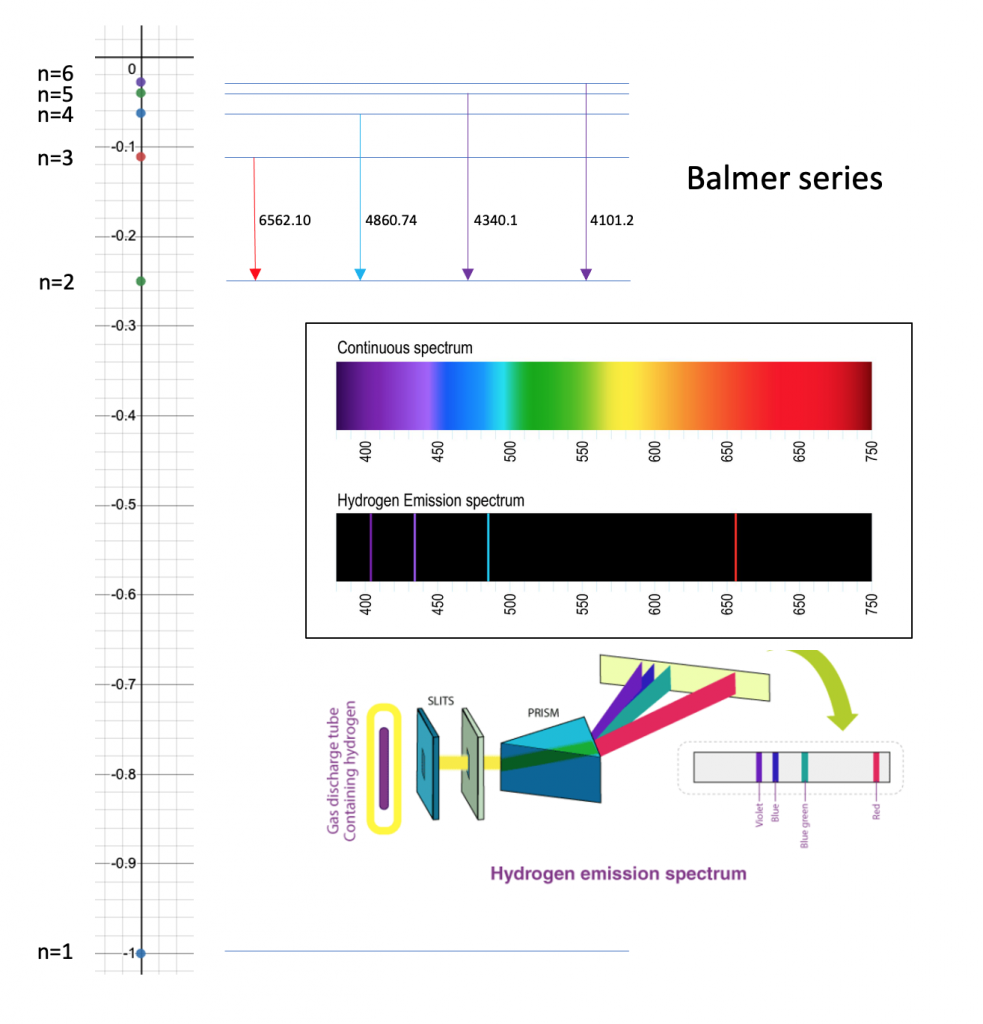
Calculate the wavelength in Angstroms of the photon that is emitted when an electron in the Bohr's orbit, n = 2 returns to the orbit, - Sarthaks eConnect | Largest Online Education Community
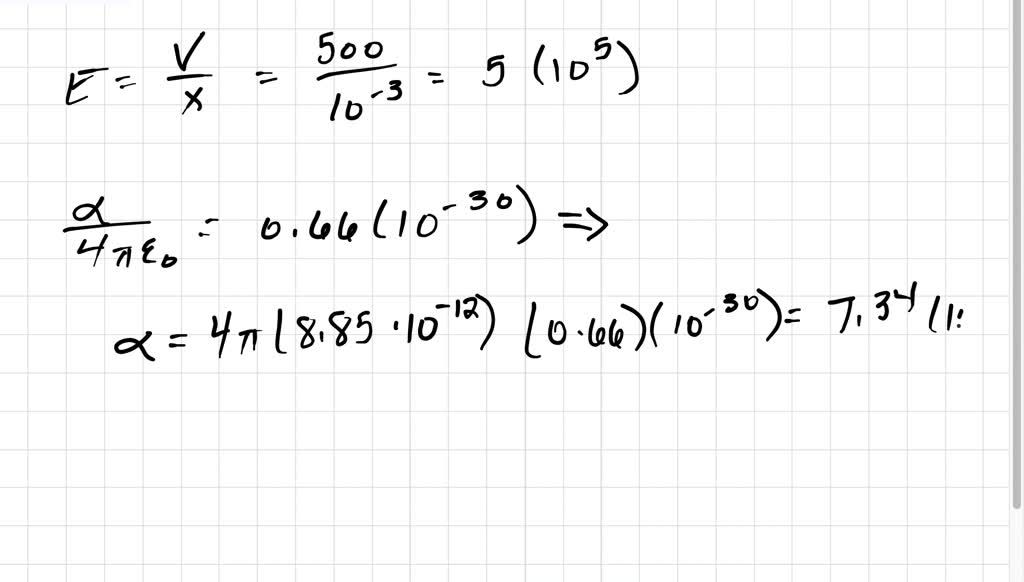
SOLVED:A hydrogen atom (with the Bohr radius of half an angstrom) is situated between two metal plates 1 mm apart, which are connected to opposite terminals of a 500 V battery. What

Calculate the wavelength in Angstroms of the photon that is emitted when an electron is Bohr orbit n = 2 return to the orbit n = 1 in the hydrogen atom .The

Calculate the wavelength in Angstroms of the photon that is emitted when an electron is Bohr orb... - YouTube

Calculate the wavelength in angstrom of the photon that is emitted when an electron in Bohr orbit n = 2 returns to the orbit n = 1 in the hydrogen atom. The